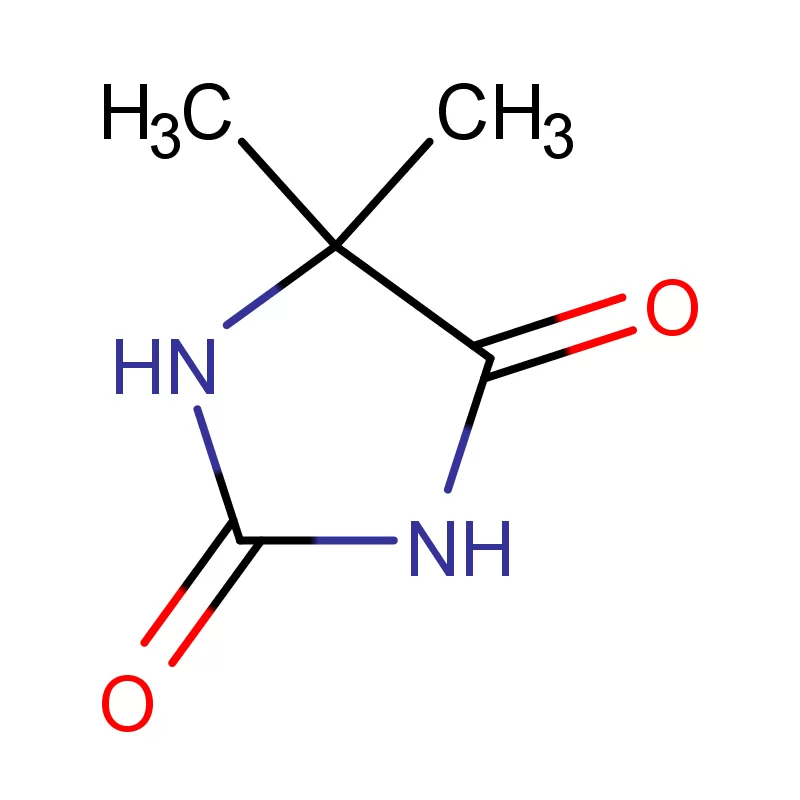
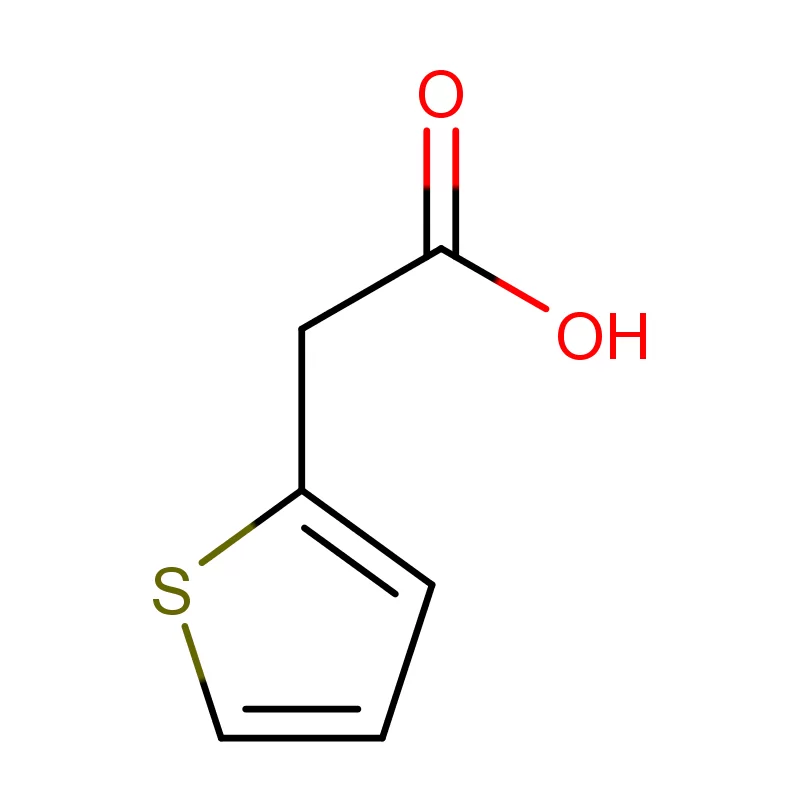
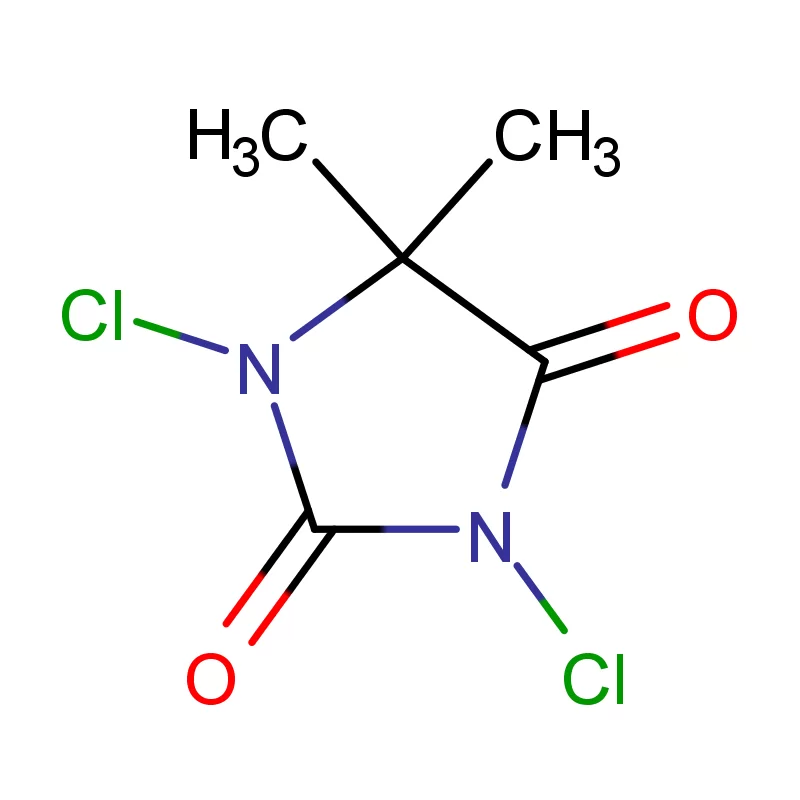
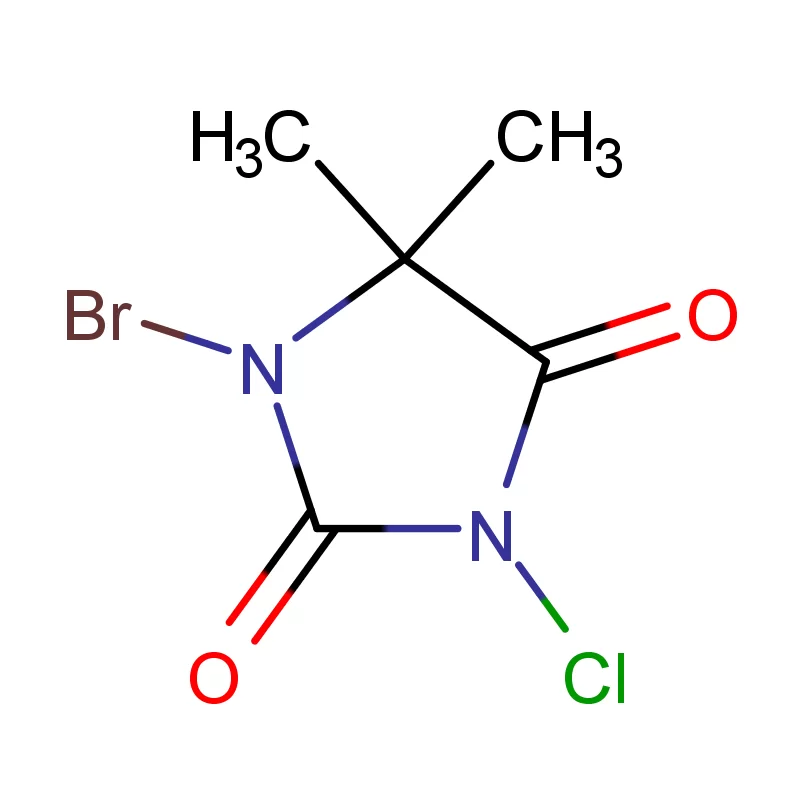
The Democratic Republic of the Congo faces significant challenges in its pharmaceutical sector, including limited local manufacturing capacity, reliance on imports, and issues with supply chain integrity. The country's vast geography and underdeveloped infrastructure contribute to logistical difficulties in distributing essential medicines. Demand for pharmaceutical intermediates is rising alongside growing healthcare needs.
Despite these hurdles, there’s a growing recognition of the importance of strengthening the local pharmaceutical industry. Government initiatives aim to encourage investment and promote local production. The availability of fine chemicals for manufacturing locally is crucial to these efforts. Investment in quality control and regulatory frameworks is also gaining traction.
The presence of abundant natural resources presents an opportunity for developing a more self-sufficient pharmaceutical supply chain. However, political and economic instability, coupled with limited skilled labor, remain significant barriers to progress. Addressing these factors is essential for realizing the DRC’s potential as a regional pharmaceutical hub. The use of industrial water treatment chemicals is important for ensuring purity during manufacturing.